 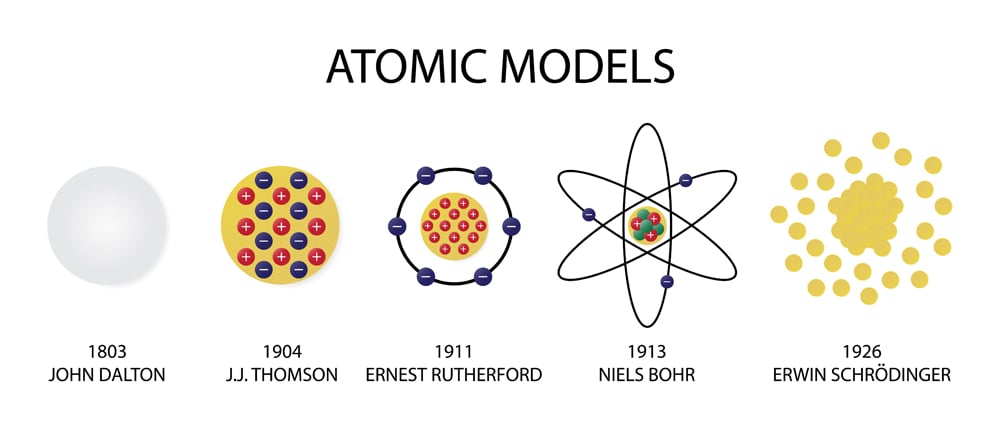 
Just as Bohr imagined that the atoms were little planets revolving around the nucleus as though it were a sun, other scientists tried to extend that idea by imagining that the little planets, just like real planets, had spin. It got named spin back when people were working with the Bohr model and trying to extend it to atoms beyond hydrogen. Spin is just a property that electrons (and other particles) have. 
For another layer, you can take the fact that you can never cool anything down to exactly 0K (-273.15C) (although you can get close) and so nothing will ever have 0 velocity. (1 value in a range of reals is like trying to throw a dart at a dartboard with an infinitely thin wire and hitting the wire). With this uncertainty, the velocity is almost definitely not 0. As we can't physically measure to perfect accuracy, there is an uncertainty in both measurements of the degree that we know it's probably stationary and it's probably 'over there'. So, if you know with 0 uncertainty what the velocity is, then you have no idea where it is, and all future involvement of the particle is pretty much irrelevant (how is the electron going to diffract around an atom if the electron is in a different galaxy?). The square (probability function) shows that it has an equal chance of being anywhere. If you take the infinite wavelength interpretation, then it would be nearly 0 (1/inf) but constant everywhere. Isn't to do with the fact that the velocity is not quite 0? if you know it is exactly 0 then the uncertainty in the position is infinite as well (momentum is a function of velocity, so delta P = 0 -> delta V = 0 -> delta X = inf) therefore it has an equal probability of being anywhere. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
